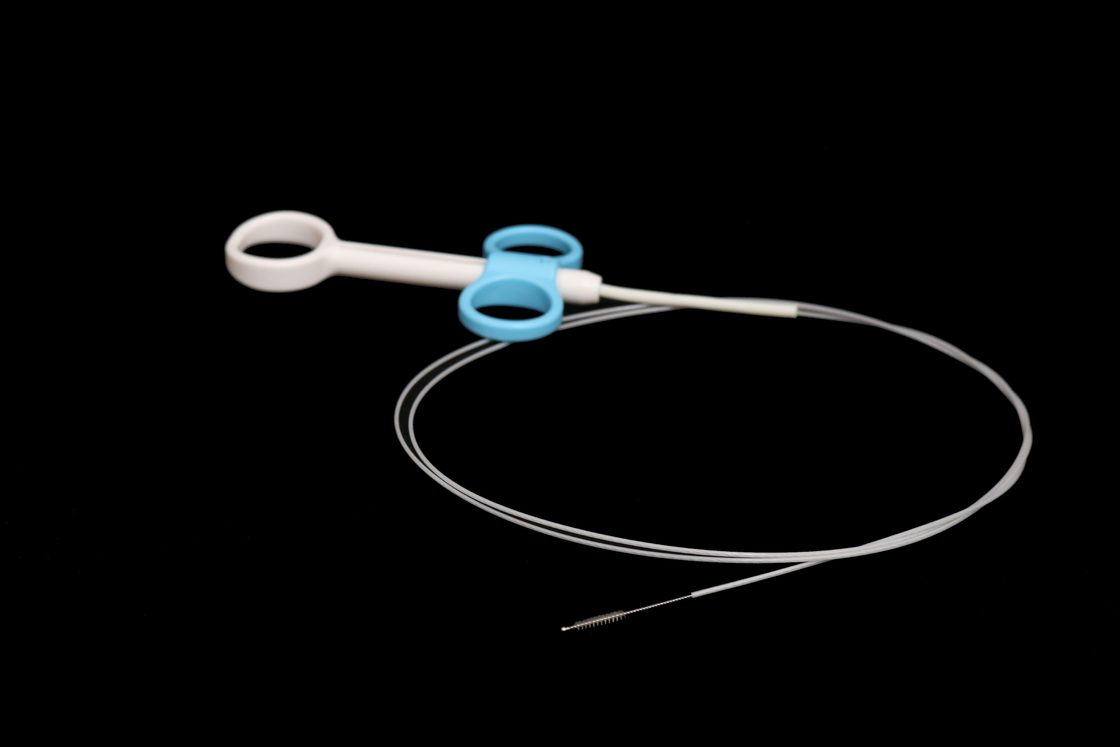
Sterilized Disposable Cell Brush With Endoscopy CE/ISO13485 Certification
-
High Light
medical cytology brush
,single use cytology brush
-
Product NameCytology Brush
-
Channel I.D≥2.0/2.8
-
CertificationCE/ISO13485
-
Main MaterialStainless Steel, ABS, Nylon, PTFE
-
ColorPink
-
Working Length1200/1600/2300mm
-
Instrument ClassificationClass II
-
PortShanghai
-
Place of OriginChina
-
Brand NameChangmei
-
CertificationCE
-
Model NumberBC
-
Minimum Order Quantity500pcs
-
Price$4~20/pcs
-
Packaging DetailsCarton size: 535*380*330mm
-
Delivery Time15-30days
-
Payment TermsT/T
-
Supply Ability50000pcs/year
Sterilized Disposable Cell Brush With Endoscopy CE/ISO13485 Certification
Medical Instruments Disposable Cytology Brush Used With Endoscopy
Intended use:
Be used for cell tissue sampling on clinical.
Features:
1. Brush of integration, without possibility of breaking off
2. With high smoothness, the outer tube is not easy to get bended
3. With suitable stiffness of bristles, it is easy for cell tissue sampling
4. Sterilized product, no need to re-sterilize before use, only for disposable use
5. Smooth sphere brush head will not hurt human tissue or endoscope channel
6. Plastic protective tube to avoid cells to fall off or leave in the endoscope channel
Specifications:
| Model | Sheath O.D. (mm) |
Channnel O.D. (mm) |
Working length (mm) |
Brush shape |
| BC-20E-A | 1.7 | ≥2.0 | 1200 | Straight Shape |
| BC-28K-A | 2.3 | ≥2.8 | 1600 | |
| BC-28U-A | 2.3 | ≥2.8 | 2300 | |
| BC-20E-B | 1.7 | ≥2.0 | 1200 | U-Shape |
| BC-28K-B | 2.3 | ≥2.8 | 1600 | |
| BC-28U-B | 2.3 | ≥2.8 | 2300 |
Packing & Delivery:
Packing:Dupont bag sterilized packaging, two layers plastic sealing, foam paper and box packaging.
Lead time:Within 15 working days after receiving 100% prepayment.
Why Choose Us
Jiangsu Changmei Medtech Co., Ltd. is mainly engaged in medical equipment products and production equipment design and development and manufacturing, in particular focus on the medical balloon catheter market, the company in the innovative balloon technology, production equipment and integrated manufacturing field closely follow the trend of the world, is committed to the medical balloon catheter in the case of domestic.
- With a modern standard workshop of 6000m2,among which,1600m2 are class 100000 clean rooms in compliance with YY0033;
- National class II and class III medical device production license;
- EN ISO 13485:2012 authentication certificate by TÜV SÜD;
- With numerous patents, Jiangsu Province Science and Technology of private enterprises;
- Experienced in interventional and endoscope equipment research and development of minimally invasive diagnosis and treatment;
- Our products have been in service for improving patients’ quality of life all over the world.
FAQ
Q: Are you trading company or manufacturer ?
A: We are manufacturer located in Changzhou Jiangsu China,which could supply OEM&ODM services.
Q: How long is your delivery time ?
A: Generally it is 7 working days if the goods are in stock. Or it is 30-60 days for regular production.
Q: Can you produce according to customers' design ?
A:Yes, we are professional manufacturer; OEM and ODM are both welcomed.
1) Silk print logo on the product;
2) Customized product housing;
3) Customized Color box;
4) Any your Idea on product we can help you to design and put it into production.
Q: Do you provide samples ? Is it free or extra ?
A: Yes, it depends on different product type whose sample policy is different.If you are interested on our product sincerely,we can give you some support.
Q: What is your terms of payment ?
A: T/T. If you have any question, Please contact with us.
![]()
![]()
![]()